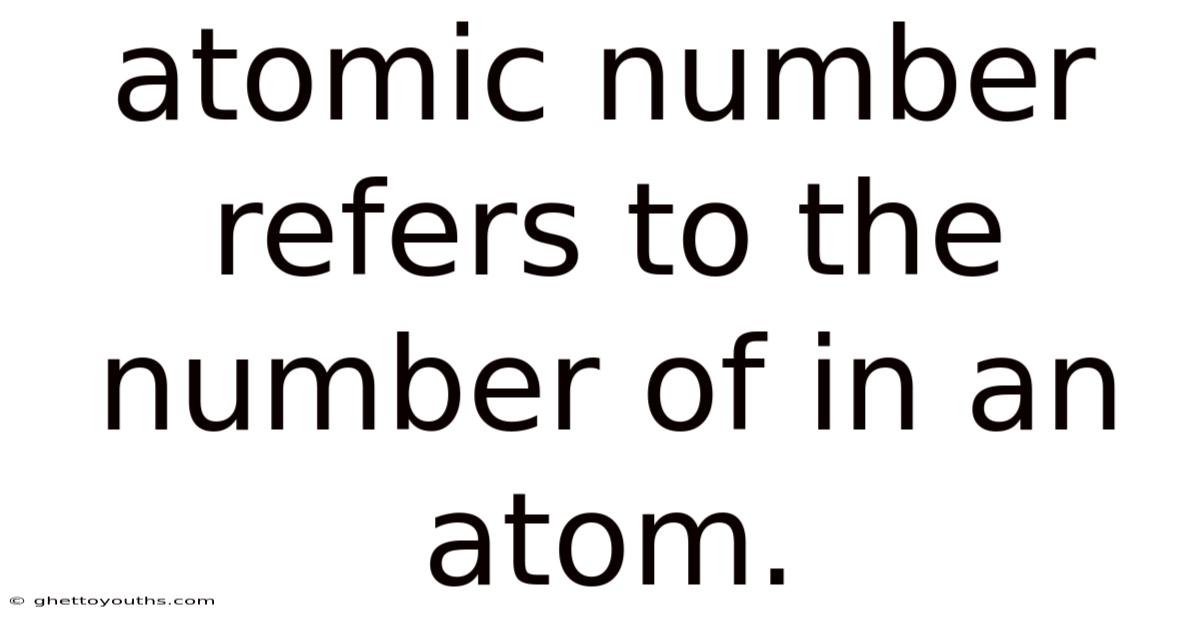The atomic number, a fundamental concept in chemistry and physics, serves as the identity card for every element in the periodic table. Day to day, it's more than just a number; it's a key to understanding an element's chemical behavior, its properties, and its place in the grand scheme of matter. This article walks through the core meaning of the atomic number, its historical context, its significance in understanding atomic structure, and its applications in various scientific fields.
Easier said than done, but still worth knowing.
Introduction
Imagine a world where every person is identified not by a name, but by a unique number that reveals everything about their origins, characteristics, and potential. In the world of atoms, that number is the atomic number. Because of that, it's a simple integer that holds the key to understanding the complex behavior of elements. In real terms, the atomic number defines an element and dictates its chemical properties. Elements with the same atomic number belong to the same element family, regardless of their other properties Worth keeping that in mind. Simple as that..
The Essence of Atomic Number
So, what exactly does the atomic number refer to? In practice, in the heart of every atom lies the nucleus, a tiny, dense region packed with particles called protons and neutrons. The atomic number is equal to the number of protons found in the nucleus of an atom. This seemingly simple definition has profound implications Took long enough..
A Journey Through History
The concept of atomic number wasn't always as clear-cut as it is today. The quest to understand the fundamental building blocks of matter and their organization has been a long and winding journey Not complicated — just consistent..
- Early Ideas: In the early 19th century, John Dalton proposed the atomic theory, suggesting that each element is composed of unique atoms. Even so, the idea of a distinct "atomic number" was yet to emerge.
- Mendeleev's Periodic Table: Dmitri Mendeleev's periodic table, published in 1869, was a notable achievement. He arranged elements in order of increasing atomic weight, noticing repeating patterns in their properties. Although atomic weight was a useful organizing principle, it wasn't the fundamental identifier we now know as the atomic number.
- Discovery of the Nucleus: Ernest Rutherford's gold foil experiment in 1911 revolutionized our understanding of the atom. He discovered that atoms have a small, dense, positively charged nucleus.
- Moseley's Contribution: Henry Moseley, in 1913, made a crucial breakthrough. By bombarding elements with electrons and measuring the wavelengths of emitted X-rays, he found a direct relationship between the wavelength and the charge of the nucleus. He realized that the square root of the X-ray frequency was proportional to the number of positive charges in the nucleus. This number, which he called the atomic number, provided a more accurate and fundamental ordering of the elements than atomic weight.
- Confirmation of Protons: It was later discovered that the positive charge in the nucleus was due to particles called protons. This discovery solidified the definition of the atomic number as the number of protons in an atom's nucleus.
Decoding Atomic Structure
The atomic number is not just a label; it's a window into the inner workings of an atom.
- Number of Protons: As already stated, the atomic number defines the number of protons in the nucleus. To give you an idea, hydrogen (H) has an atomic number of 1, meaning it has one proton. Oxygen (O) has an atomic number of 8, meaning it has eight protons.
- Neutral Atoms: In a neutral atom, the number of protons is equal to the number of electrons orbiting the nucleus. This balance ensures that the atom has no overall electrical charge. That's why, the atomic number also tells us the number of electrons in a neutral atom.
- Chemical Identity: The number of protons dictates the chemical properties of an element. Atoms with the same number of protons behave similarly in chemical reactions. Take this: all atoms with one proton are hydrogen atoms, and they will all exhibit the characteristic properties of hydrogen.
- Isotopes: While the number of protons defines an element, the number of neutrons in the nucleus can vary. Atoms of the same element with different numbers of neutrons are called isotopes. Isotopes have the same atomic number but different mass numbers (the total number of protons and neutrons). Here's a good example: carbon-12, carbon-13, and carbon-14 are all isotopes of carbon. They all have six protons (atomic number 6), but they have 6, 7, and 8 neutrons, respectively.
- Ions: Atoms can also gain or lose electrons, forming ions. Ions are atoms with an electrical charge. If an atom loses electrons, it becomes a positive ion (cation). If it gains electrons, it becomes a negative ion (anion). The atomic number remains the same, as the number of protons doesn't change.
The Periodic Table: A Masterpiece of Organization
The periodic table is a testament to the power of the atomic number. It organizes elements in order of increasing atomic number, arranging them in rows (periods) and columns (groups) based on their similar chemical properties.
- Periods: Elements in the same period have the same number of electron shells. As you move across a period, the atomic number increases, and the properties of the elements change gradually.
- Groups: Elements in the same group have the same number of valence electrons (electrons in the outermost shell). Valence electrons determine how an element interacts with other elements. Elements in the same group tend to exhibit similar chemical behavior.
- Organization by Atomic Number: The periodic table's organization by atomic number, rather than atomic weight, resolved inconsistencies that arose when Mendeleev used atomic weight. Here's one way to look at it: tellurium (Te) has a higher atomic weight than iodine (I), but placing them in order of atomic weight would have put them in the wrong groups based on their chemical properties. Moseley's discovery and the use of atomic number corrected this anomaly.
Beyond the Basics: Applications and Implications
The atomic number is more than just a theoretical concept; it has numerous practical applications and implications across various scientific fields.
- Nuclear Chemistry: The atomic number is crucial in nuclear chemistry, which deals with the structure, properties, and reactions of atomic nuclei. Nuclear reactions involve changes in the number of protons and neutrons in the nucleus, leading to the formation of different elements or isotopes.
- Materials Science: The properties of materials are directly related to the elements they contain and their atomic structure. The atomic number helps predict and understand the behavior of materials, leading to the development of new materials with desired properties.
- Spectroscopy: Spectroscopic techniques analyze the interaction of matter with electromagnetic radiation. Each element has a unique spectral signature based on its atomic number and electron configuration. Spectroscopy is used to identify elements in various samples, from stars to pollutants.
- Medical Imaging: Radioactive isotopes, which are characterized by their atomic number and mass number, are used in medical imaging techniques such as PET scans and SPECT scans. These techniques allow doctors to visualize internal organs and detect diseases.
- Forensic Science: The atomic number is used in forensic science to identify trace elements in samples found at crime scenes. This can help link suspects to crimes or identify the origin of unknown substances.
- Quantum Mechanics: The atomic number is a fundamental parameter in quantum mechanical calculations of atomic structure and properties. The Schrödinger equation, which describes the behavior of electrons in atoms, requires the atomic number as input.
- Cosmology: The abundance of elements in the universe is determined by nuclear reactions that occurred in the early universe and in stars. The atomic number is essential for understanding these processes and the evolution of the universe.
The Significance of Isotopes
As mentioned earlier, isotopes are atoms of the same element with different numbers of neutrons. While isotopes share the same atomic number and therefore the same chemical properties, their different mass numbers lead to variations in their physical properties and nuclear behavior.
- Stable Isotopes: Some isotopes are stable, meaning their nuclei do not spontaneously decay. These isotopes are used in various applications, such as isotope tracing and nuclear magnetic resonance (NMR) spectroscopy.
- Radioactive Isotopes: Other isotopes are radioactive, meaning their nuclei are unstable and decay over time, emitting particles and energy. Radioactive isotopes are used in medical imaging, cancer therapy, carbon dating, and other applications.
- Isotope Effects: The different masses of isotopes can lead to small variations in their reaction rates and equilibrium constants. These isotope effects are used to study reaction mechanisms and to separate isotopes.
Unveiling New Elements
The periodic table is not a closed book. Which means scientists continue to discover and synthesize new elements, extending the table to higher atomic numbers. These synthetic elements are typically produced in particle accelerators by bombarding heavy nuclei with other nuclei That's the part that actually makes a difference..
- Transuranic Elements: Elements with atomic numbers greater than 92 (the atomic number of uranium) are called transuranic elements. These elements are all synthetic and radioactive.
- Naming New Elements: When a new element is discovered, it is initially given a temporary name and symbol. The discoverers then have the opportunity to propose a permanent name, which is subject to approval by the International Union of Pure and Applied Chemistry (IUPAC).
- Island of Stability: Scientists are searching for an "island of stability" in the sea of radioactive transuranic elements. This hypothetical region of the periodic table would contain superheavy elements with relatively long half-lives, potentially opening up new possibilities for materials science and other fields.
Tren & Perkembangan Terkini
The exploration of atomic numbers and the elements they represent continues to evolve with ongoing research and technological advancements. Here's a glimpse into the current trends and developments:
- Superheavy Element Synthesis: Scientists are relentlessly pursuing the synthesis of new superheavy elements with even higher atomic numbers. This involves pushing the boundaries of accelerator technology and nuclear reaction theory.
- Isotope Production and Applications: The production of specific isotopes for medical, industrial, and research applications is a growing field. New techniques are being developed to improve isotope production efficiency and purity.
- Advanced Spectroscopic Techniques: Advanced spectroscopic techniques are being developed to probe the electronic structure and properties of elements and isotopes with unprecedented precision.
- Quantum Computing for Atomic Calculations: Quantum computers hold the promise of performing highly accurate calculations of atomic structure and properties, which are currently intractable for classical computers.
- Data-Driven Materials Discovery: Machine learning and artificial intelligence are being used to analyze vast datasets of elemental properties and predict new materials with desired characteristics.
Tips & Expert Advice
Understanding the atomic number and its significance can be greatly enhanced by following these tips and expert advice:
- Visualize Atomic Structure: Use diagrams and models to visualize the arrangement of protons, neutrons, and electrons in atoms. This will help you understand the relationship between the atomic number and the overall structure of an atom.
- Explore the Periodic Table: Spend time exploring the periodic table and familiarizing yourself with the properties of different elements. Pay attention to the trends in atomic number, atomic mass, electronegativity, ionization energy, and other properties.
- Practice with Examples: Work through examples of how to determine the number of protons, neutrons, and electrons in atoms, ions, and isotopes. This will solidify your understanding of the concepts.
- Stay Curious: Read articles, watch videos, and attend lectures on atomic structure and the periodic table. Science is a constantly evolving field, so there's always something new to learn.
- Use Online Resources: Take advantage of the many online resources available, such as interactive periodic tables, simulations of atomic structure, and educational videos.
- Connect with Experts: Join online forums or communities where you can ask questions and discuss topics related to atomic structure and chemistry with experts and other students.
- Apply Your Knowledge: Look for opportunities to apply your knowledge of atomic numbers and elemental properties to real-world problems, such as understanding the composition of materials or interpreting scientific data.
FAQ (Frequently Asked Questions)
-
Q: What is the atomic number?
- A: The atomic number is the number of protons in the nucleus of an atom.
-
Q: Why is the atomic number important?
- A: The atomic number defines an element and dictates its chemical properties.
-
Q: How is the atomic number related to the periodic table?
- A: The periodic table is organized in order of increasing atomic number.
-
Q: What are isotopes?
- A: Isotopes are atoms of the same element with different numbers of neutrons.
-
Q: How are new elements discovered?
- A: New elements are typically synthesized in particle accelerators by bombarding heavy nuclei with other nuclei.
-
Q: Can the atomic number change?
- A: The atomic number can only change in nuclear reactions. In chemical reactions, the atomic number remains the same.
-
Q: Is the atomic number a whole number?
- A: Yes, the atomic number is always a whole number because you can't have a fraction of a proton.
Conclusion
The atomic number is a cornerstone of our understanding of matter. Worth adding: from its historical roots in the quest to understand the elements to its modern applications in diverse scientific fields, the atomic number continues to be a vital tool for scientists and researchers. Think about it: it's a reminder that even the most complex phenomena can be understood through simple, fundamental principles. It's more than just a number; it's the key to unlocking the secrets of the universe That's the part that actually makes a difference..
So, how does this new understanding of the atomic number change your perception of the world around you? Are you inspired to delve deeper into the mysteries of chemistry and physics?