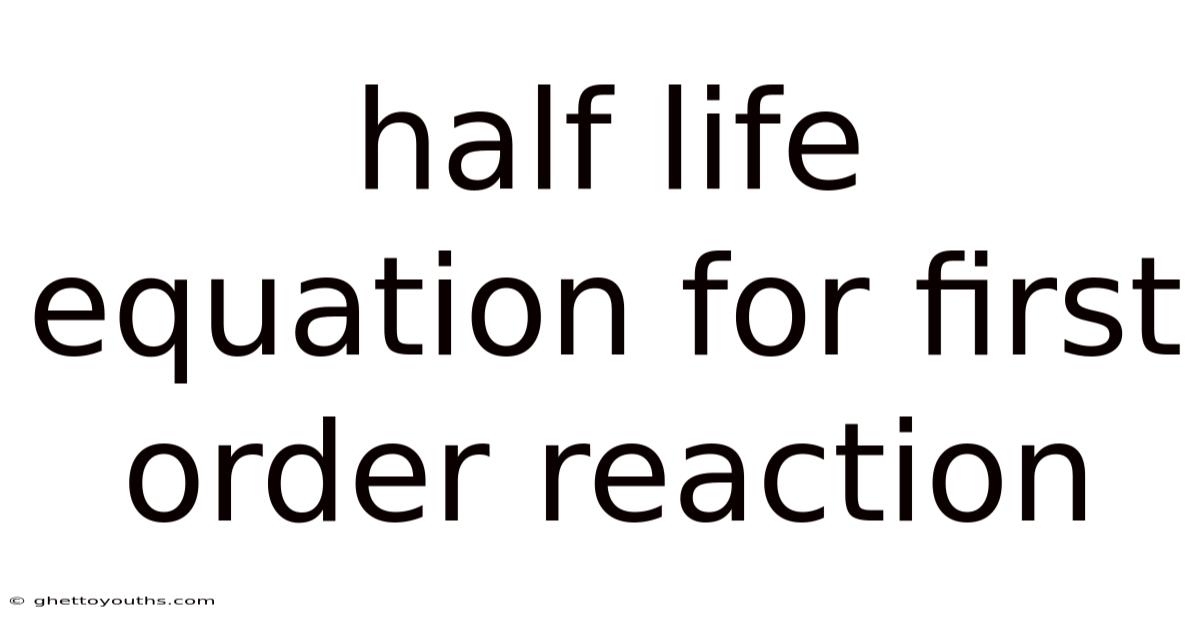Let's dive into the fascinating world of chemical kinetics and explore a fundamental concept: the half-life equation for first-order reactions. Whether you're a student grappling with chemistry concepts or simply curious about how reactions proceed over time, understanding half-life is crucial. This article will provide a comprehensive breakdown, from the basic definition to practical applications and common misconceptions.
Introduction
Imagine you have a radioactive substance decaying, or a medication breaking down in your bloodstream. In both cases, the rate at which these processes occur isn't constant. Now, instead, they follow a predictable pattern, especially if they are first-order reactions. This is where the concept of half-life comes in – it's the time it takes for half of the initial amount of a reactant to be consumed in a chemical reaction. More specifically, we’re focusing on the half-life equation for first-order reactions, a common type of reaction found everywhere from nuclear decay to enzyme kinetics That's the part that actually makes a difference..
People argue about this. Here's where I land on it Small thing, real impact..
Understanding half-life not only helps us predict how long a reaction will take but also allows us to manage resources, estimate the age of ancient artifacts, and even design better medications. Let's embark on this journey to unravel the complexities and practical applications of half-life in first-order reactions.
At its core, where a lot of people lose the thread.
Half-Life: The Basics
The half-life (( t_{1/2} )) is a term that signifies the time required for a quantity to reduce to half of its initial value. In the context of chemical kinetics, it specifically refers to the duration it takes for the concentration of a reactant to decrease to half of its original concentration. This concept is particularly relevant for first-order reactions, which exhibit a consistent and predictable rate of decay.
A first-order reaction is one in which the reaction rate depends linearly on the concentration of only one reactant. What this tells us is as the concentration of the reactant decreases, the reaction rate also decreases proportionally. A classic example is the radioactive decay of certain isotopes Simple as that..
Comprehensive Overview of First-Order Reactions
To truly grasp the significance of the half-life equation, we need to understand the characteristics of first-order reactions. Let's delve deeper into the definition, rate law, and integrated rate law for first-order reactions.
Definition and Characteristics
A first-order reaction is characterized by the fact that its rate depends only on the concentration of one reactant. Mathematically, this can be expressed through the rate law:
[ \text{Rate} = -\frac{d[A]}{dt} = k[A] ]
Where:
- ( [A] ) is the concentration of the reactant. But * ( t ) is time. Plus, * ( k ) is the rate constant, which is specific to the reaction and temperature. * ( -\frac{d[A]}{dt} ) represents the rate of decrease of the reactant concentration with respect to time.
This equation tells us that the rate of the reaction is directly proportional to the concentration of reactant A.
Integrated Rate Law
The integrated rate law for a first-order reaction is derived from the differential rate law through calculus. It relates the concentration of the reactant to time, allowing us to predict the concentration at any point in the reaction. The integrated rate law is expressed as:
[ \ln[A]_t - \ln[A]_0 = -kt ]
Where:
- ( [A]_t ) is the concentration of reactant A at time ( t ). Because of that, * ( [A]_0 ) is the initial concentration of reactant A at time ( t = 0 ). * ( k ) is the rate constant.
This equation can also be written in the exponential form as:
[ [A]_t = [A]_0 e^{-kt} ]
This exponential decay is a hallmark of first-order reactions, signifying that the concentration of the reactant decreases exponentially over time Simple as that..
Graphical Representation
Graphically, a first-order reaction can be represented in two primary ways:
- Concentration vs. Time: A plot of ( [A]_t ) against ( t ) yields a decaying exponential curve.
- Natural Logarithm of Concentration vs. Time: A plot of ( \ln[A]_t ) against ( t ) yields a straight line with a slope of ( -k ). This linear relationship makes it easy to determine the rate constant ( k ) experimentally.
Derivation of the Half-Life Equation
Now that we understand the basics of first-order reactions, let's derive the half-life equation. The half-life (( t_{1/2} )) is the time it takes for the initial concentration of the reactant to reduce to half, i.e., ( [A]_t = \frac{1}{2}[A]_0 ).
Honestly, this part trips people up more than it should.
Using the integrated rate law:
[ \ln[A]_t - \ln[A]_0 = -kt ]
Substitute ( [A]_t = \frac{1}{2}[A]_0 ):
[ \ln\left(\frac{1}{2}[A]_0\right) - \ln[A]0 = -kt{1/2} ]
Using logarithmic properties, we can simplify this:
[ \ln\left(\frac{1}{2}\right) + \ln[A]_0 - \ln[A]0 = -kt{1/2} ]
[ \ln\left(\frac{1}{2}\right) = -kt_{1/2} ]
Since ( \ln\left(\frac{1}{2}\right) = -\ln(2) ), we have:
[ -\ln(2) = -kt_{1/2} ]
Solving for ( t_{1/2} ):
[ t_{1/2} = \frac{\ln(2)}{k} ]
Which means, the half-life equation for a first-order reaction is:
[ t_{1/2} = \frac{0.693}{k} ]
Where ( \ln(2) \approx 0.693 ).
Key Points from the Derivation
- The half-life is independent of the initial concentration ( [A]_0 ). This is a unique characteristic of first-order reactions.
- The half-life is inversely proportional to the rate constant ( k ). A larger rate constant means a shorter half-life, indicating a faster reaction.
Practical Applications of the Half-Life Equation
The half-life equation isn’t just a theoretical concept; it has numerous real-world applications across various fields.
1. Radioactive Decay
One of the most well-known applications is in the field of nuclear chemistry and physics. Radioactive isotopes decay via first-order kinetics. The half-life of a radioactive isotope is the time it takes for half of the atoms in a sample to decay.
People argue about this. Here's where I land on it The details matter here..
- Radiocarbon Dating: Archaeologists use the half-life of carbon-14 (approximately 5,730 years) to determine the age of organic materials. By measuring the amount of carbon-14 remaining in a sample, they can estimate when the organism died.
- Medical Imaging: Radioactive isotopes like technetium-99m are used in medical imaging to diagnose various conditions. Knowing the half-life helps in determining the optimal dosage and timing of imaging procedures.
- Nuclear Waste Management: Understanding the half-lives of different radioactive isotopes is essential for safely storing nuclear waste. Isotopes with long half-lives require long-term storage solutions.
2. Pharmacokinetics
In pharmacology, the half-life of a drug is the time it takes for the concentration of the drug in the plasma or blood to reduce by half. This is a critical parameter in determining:
- Dosage Regimen: The half-life helps determine how frequently a drug should be administered to maintain therapeutic levels in the body. Drugs with short half-lives need to be taken more frequently.
- Drug Clearance: The half-life is related to the clearance rate of a drug from the body. Drugs with longer half-lives take longer to be eliminated.
- Steady-State Concentration: Understanding half-life helps predict how long it will take for a drug to reach a steady-state concentration in the body, where the rate of drug administration equals the rate of drug elimination.
3. Chemical Kinetics in Industry
In industrial chemistry, many reactions follow first-order kinetics, particularly decomposition reactions. Understanding the half-life can help in:
- Process Optimization: Predicting how long it will take for a reactant to degrade or a product to form.
- Quality Control: Monitoring the stability of chemicals and ensuring they meet required specifications over time.
- Reaction Engineering: Designing reactors and optimizing reaction conditions based on reaction rates and half-lives.
4. Environmental Science
The degradation of pollutants in the environment often follows first-order kinetics. Knowing the half-life of a pollutant can help in:
- Risk Assessment: Estimating how long a pollutant will persist in the environment.
- Remediation Strategies: Planning remediation efforts based on the natural decay rate of the pollutant.
- Regulation and Policy: Setting environmental standards and regulations based on the persistence of pollutants.
Common Misconceptions and Pitfalls
While the half-life equation is straightforward, several misconceptions can lead to errors in its application:
- Misconception 1: Half-Life Depends on Initial Concentration: For first-order reactions, the half-life is independent of the initial concentration. This is a crucial point that distinguishes it from other reaction orders.
- Misconception 2: All Reactions Have a Half-Life: Only reactions that follow first-order kinetics (or can be approximated as such under certain conditions) have a constant half-life. Higher-order reactions have half-lives that depend on the concentration of reactants.
- Pitfall 1: Incorrectly Identifying First-Order Reactions: It's essential to confirm that a reaction is indeed first-order before applying the half-life equation. This can be done experimentally by analyzing rate data.
- Pitfall 2: Forgetting Units: Always confirm that the units of the rate constant ( k ) and time are consistent to avoid errors in calculations. If ( k ) is in ( \text{s}^{-1} ), the half-life will be in seconds.
- Pitfall 3: Assuming Constant Temperature: The rate constant ( k ) (and therefore the half-life) is temperature-dependent. Calculations are valid only if the temperature is constant.
Examples and Problem-Solving
Let's work through some examples to illustrate how the half-life equation is applied in practice Took long enough..
Example 1: Radioactive Decay
The half-life of iodine-131 is 8.02 days. If you start with a 100 mg sample, how much will remain after 24.06 days?
- Find the Rate Constant ( k ): [ k = \frac{0.693}{t_{1/2}} = \frac{0.693}{8.02 \text{ days}} \approx 0.0864 \text{ days}^{-1} ]
- Use the Integrated Rate Law: [ [A]_t = [A]_0 e^{-kt} ] [ [A]_t = 100 \text{ mg} \cdot e^{-(0.0864 \text{ days}^{-1} \cdot 24.06 \text{ days})} ] [ [A]_t \approx 100 \text{ mg} \cdot e^{-2.078} \approx 100 \text{ mg} \cdot 0.125 \approx 12.5 \text{ mg} ]
After 24.06 days, approximately 12.5 mg of iodine-131 will remain.
Example 2: Drug Elimination
A drug has a half-life of 4 hours. If the initial concentration in the blood is 200 mg/L, how long will it take for the concentration to drop to 25 mg/L?
- Find the Rate Constant ( k ): [ k = \frac{0.693}{t_{1/2}} = \frac{0.693}{4 \text{ hours}} \approx 0.173 \text{ hours}^{-1} ]
- Use the Integrated Rate Law: [ \ln[A]_t - \ln[A]_0 = -kt ] [ \ln(25) - \ln(200) = -0.173 \cdot t ] [ \ln\left(\frac{25}{200}\right) = -0.173 \cdot t ] [ \ln(0.125) = -0.173 \cdot t ] [ -2.079 = -0.173 \cdot t ] [ t \approx \frac{-2.079}{-0.173} \approx 12 \text{ hours} ]
It will take approximately 12 hours for the drug concentration to drop to 25 mg/L.
The Rate Constant (k) and its Significance
The rate constant (k) is critical in understanding the speed of a reaction. Which means for a first-order reaction, the rate constant reflects the probability that a molecule will react per unit time. A larger k means the reaction proceeds faster, and vice versa.
It sounds simple, but the gap is usually here Simple, but easy to overlook..
[ k = A e^{-\frac{E_a}{RT}} ]
Where:
- A is the pre-exponential factor.
- Ea is the activation energy.
- R is the gas constant.
- T is the absolute temperature.
This equation tells us that as temperature increases, the rate constant increases exponentially, leading to a faster reaction.
Tren & Perkembangan Terbaru
- Advancements in Radiopharmaceuticals: The development of radiopharmaceuticals with shorter half-lives allows for better imaging with reduced radiation exposure to patients.
- Drug Delivery Systems: Novel drug delivery systems are being designed to control drug release and maintain therapeutic levels for longer periods, optimizing the effects and minimizing side effects.
- Environmental Remediation Techniques: Innovations in using microorganisms to degrade pollutants are leading to more effective and sustainable environmental cleanup strategies.
Tips & Expert Advice
- Always Verify Reaction Order: Before applying the half-life equation, make sure the reaction is indeed first-order.
- Pay Attention to Units: Ensure consistency in units of time and concentration throughout your calculations.
- Consider Temperature Effects: Remember that the rate constant and half-life are temperature-dependent. Use the Arrhenius equation if temperature variations are significant.
- make use of Graphical Analysis: Plotting the natural logarithm of concentration against time can help visually confirm if a reaction is first-order and determine the rate constant.
FAQ (Frequently Asked Questions)
Q: Can the half-life equation be used for second-order reactions? A: No, the half-life equation ( t_{1/2} = \frac{0.693}{k} ) is specific to first-order reactions. Second-order reactions have a different half-life equation that depends on the initial concentration of the reactant.
Q: What does a short half-life indicate about a substance? A: A short half-life indicates that the substance decays or reacts quickly. It means that half of the initial amount of the substance will be consumed in a relatively short period That's the part that actually makes a difference. Nothing fancy..
Q: How is half-life used in medicine? A: In medicine, half-life is used to determine the dosage frequency of drugs. Drugs with shorter half-lives need to be administered more frequently to maintain therapeutic levels Surprisingly effective..
Q: Is half-life affected by external factors other than temperature? A: Generally, no. Half-life is primarily affected by temperature because the rate constant, ( k ), is temperature-dependent. Other factors such as pressure and catalysts can affect the reaction rate but don't directly change the half-life of a first-order reaction.
Conclusion
Understanding the half-life equation for first-order reactions is fundamental in various fields, from chemistry and physics to pharmacology and environmental science. Its importance lies in its ability to predict reaction rates, manage resources, and optimize processes. By grasping the nuances of first-order kinetics and avoiding common misconceptions, you can effectively apply this concept to solve real-world problems.
So, how do you plan to use this knowledge in your field? Are you ready to explore the impact of half-life in your experiments or studies? Embrace this tool, and you’ll find yourself better equipped to understand and control the dynamics of chemical reactions That's the whole idea..